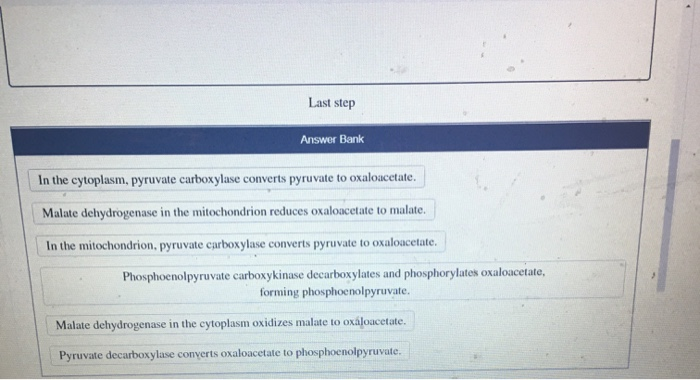
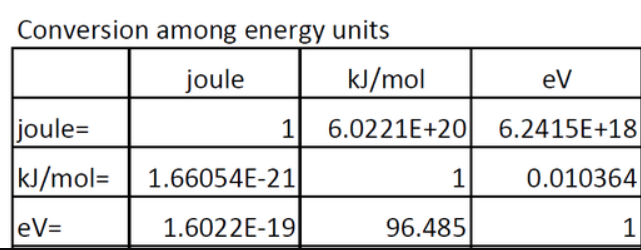
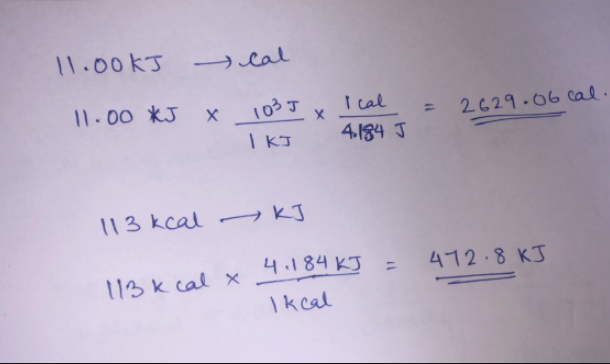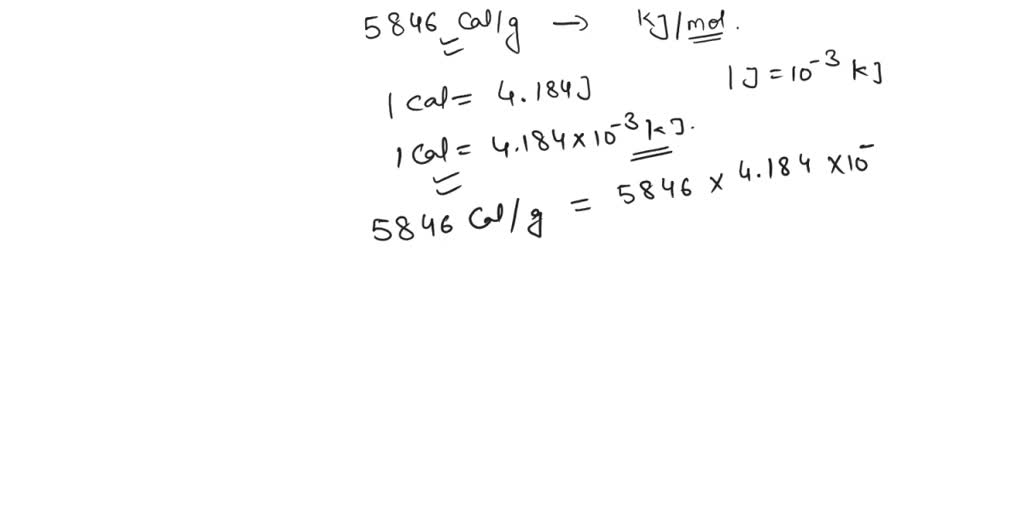The value of universal gas constant is R=8.3 J/kcal/mol. The value of R is atmosphere litre per kelvin/mol is?

SOLVED: Convert these two atomic units (a.u.) into kJ/mol by using 2625 kJ/ mol = 1 a.u. -78.7939 kJ/mol -78.7838378 kJ/mol Please show the work.
Is Kd (the dissociation constant) for a given protein-ligand pair directly correlated to binding affinity (kcal/mol, Rosetta Energy Units, etc.)? - Quora