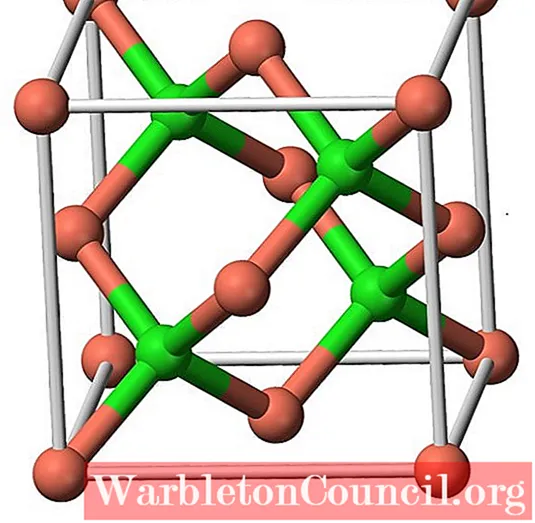
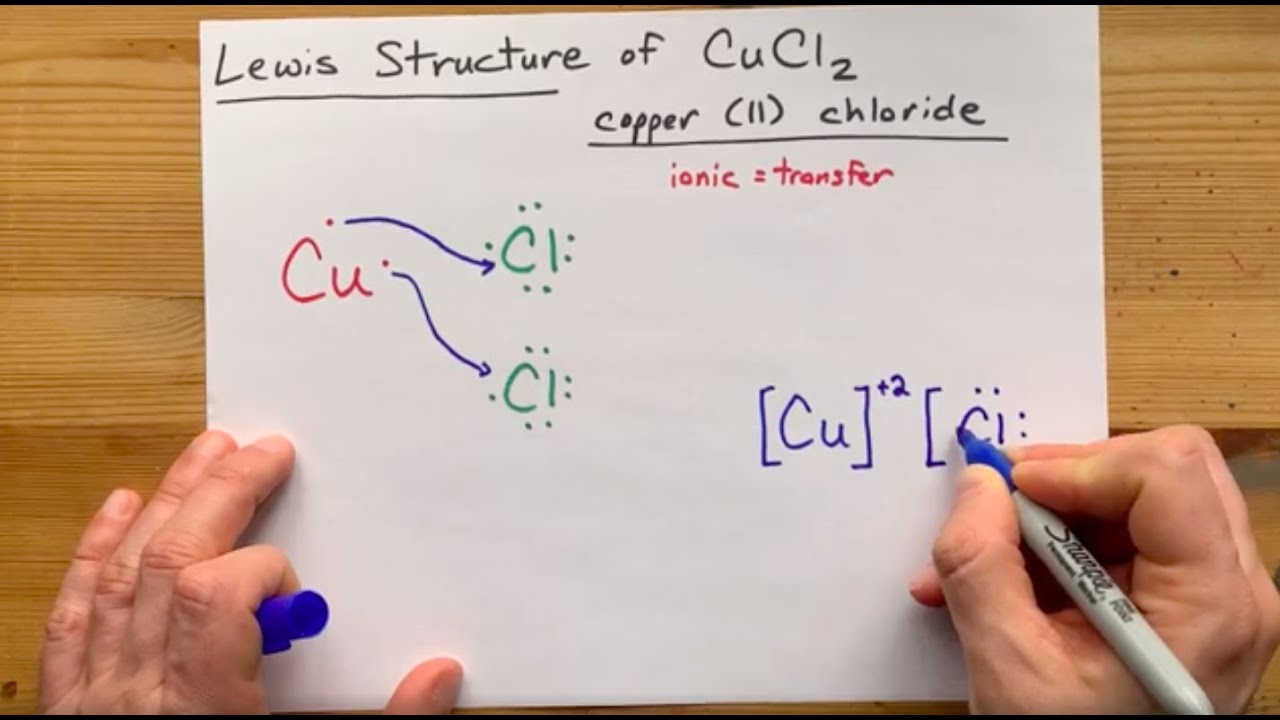
SOLVED: Determination of the empirical formula The formula for the copper chloride is one of the five shown below. Calculate the percent composition of the compound. Formula %Cu "CI CuCl CuCl2 CuCl3
![SOLVED: Formula Name a) Na3[Co(CO3)3] sodium tricarbonatocobaltate(lll) b) Rb[FeF6] rubidium hexafluoroferrate(lll) c) [CoClBr(NH3)4] tetraamminebromochlorocobalt(ll) iodide d) K[Fe(CN)6] potassium hexacyanoferrate(lll) e) Na[CuCl3] sodium ... SOLVED: Formula Name a) Na3[Co(CO3)3] sodium tricarbonatocobaltate(lll) b) Rb[FeF6] rubidium hexafluoroferrate(lll) c) [CoClBr(NH3)4] tetraamminebromochlorocobalt(ll) iodide d) K[Fe(CN)6] potassium hexacyanoferrate(lll) e) Na[CuCl3] sodium ...](https://cdn.numerade.com/ask_images/1fdbed11e2934fb3a27ece44790ace3c.jpg)
SOLVED: Formula Name a) Na3[Co(CO3)3] sodium tricarbonatocobaltate(lll) b) Rb[FeF6] rubidium hexafluoroferrate(lll) c) [CoClBr(NH3)4] tetraamminebromochlorocobalt(ll) iodide d) K[Fe(CN)6] potassium hexacyanoferrate(lll) e) Na[CuCl3] sodium ...
![SOLVED: Formula Name a) Na3[Co(CO3)3] sodium tricarbonatocobaltate(lll) b) Rb[FeF6] rubidium hexafluoroferrate(lll) c) [CoClBr(NH3)4] tetraamminebromochlorocobalt(ll) iodide d) K[Fe(CN)6] potassium hexacyanoferrate(lll) e) Na[CuCl3] sodium ... SOLVED: Formula Name a) Na3[Co(CO3)3] sodium tricarbonatocobaltate(lll) b) Rb[FeF6] rubidium hexafluoroferrate(lll) c) [CoClBr(NH3)4] tetraamminebromochlorocobalt(ll) iodide d) K[Fe(CN)6] potassium hexacyanoferrate(lll) e) Na[CuCl3] sodium ...](https://cdn.numerade.com/ask_previews/1750b7d7-5a10-4266-9512-b4561ad0c79c_large.jpg)
SOLVED: Formula Name a) Na3[Co(CO3)3] sodium tricarbonatocobaltate(lll) b) Rb[FeF6] rubidium hexafluoroferrate(lll) c) [CoClBr(NH3)4] tetraamminebromochlorocobalt(ll) iodide d) K[Fe(CN)6] potassium hexacyanoferrate(lll) e) Na[CuCl3] sodium ...
![SOLVED: Formula Name a) Na3[Co(CO3)3] sodium tricarbonatocobaltate(lll) b) Rb[FeF6] rubidium hexafluoroferrate(lll) c) [CoClBr(NH3)4] tetraamminebromochlorocobalt(ll) iodide d) K[Fe(CN)6] potassium hexacyanoferrate(lll) e) Na[CuCl3] sodium ... SOLVED: Formula Name a) Na3[Co(CO3)3] sodium tricarbonatocobaltate(lll) b) Rb[FeF6] rubidium hexafluoroferrate(lll) c) [CoClBr(NH3)4] tetraamminebromochlorocobalt(ll) iodide d) K[Fe(CN)6] potassium hexacyanoferrate(lll) e) Na[CuCl3] sodium ...](https://cdn.numerade.com/ask_previews/3ac08f8-e66f-dd-b33c-3c46c11b4d2.gif)
SOLVED: Formula Name a) Na3[Co(CO3)3] sodium tricarbonatocobaltate(lll) b) Rb[FeF6] rubidium hexafluoroferrate(lll) c) [CoClBr(NH3)4] tetraamminebromochlorocobalt(ll) iodide d) K[Fe(CN)6] potassium hexacyanoferrate(lll) e) Na[CuCl3] sodium ...

Do Now Name the following compounds – KCl – Na 2 O – CuCl 3 Write the formulas for the following compounds – Magnesium chloride – Potassium sulfate – Tin. - ppt download

Using Chemical Formulas 7-3 Beaker Breaker 1.Name the following compounds: CaCl 2 = ______ P 3 O 6 = ______ FeCl 2 = H 2 SO. - ppt download

A classmate tells you that the following compound has the name 3-propylhexane. a. Is he right? If not, what error did he make and what is the correct name? b. How could

Do Now Name the following compounds – KCl – Na 2 O – CuCl 3 Write the formulas for the following compounds – Magnesium chloride – Potassium sulfate – Tin. - ppt download