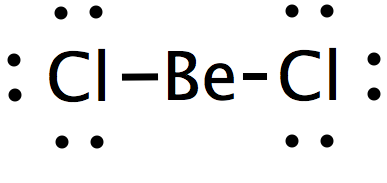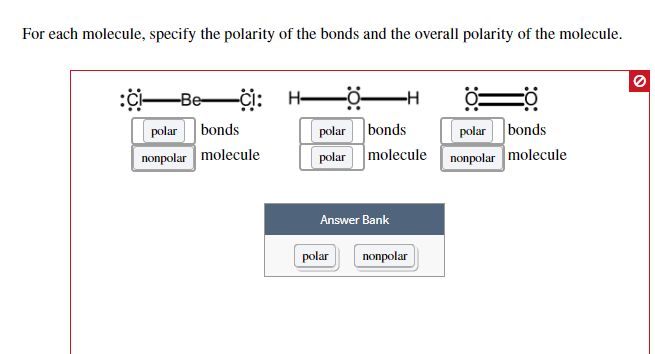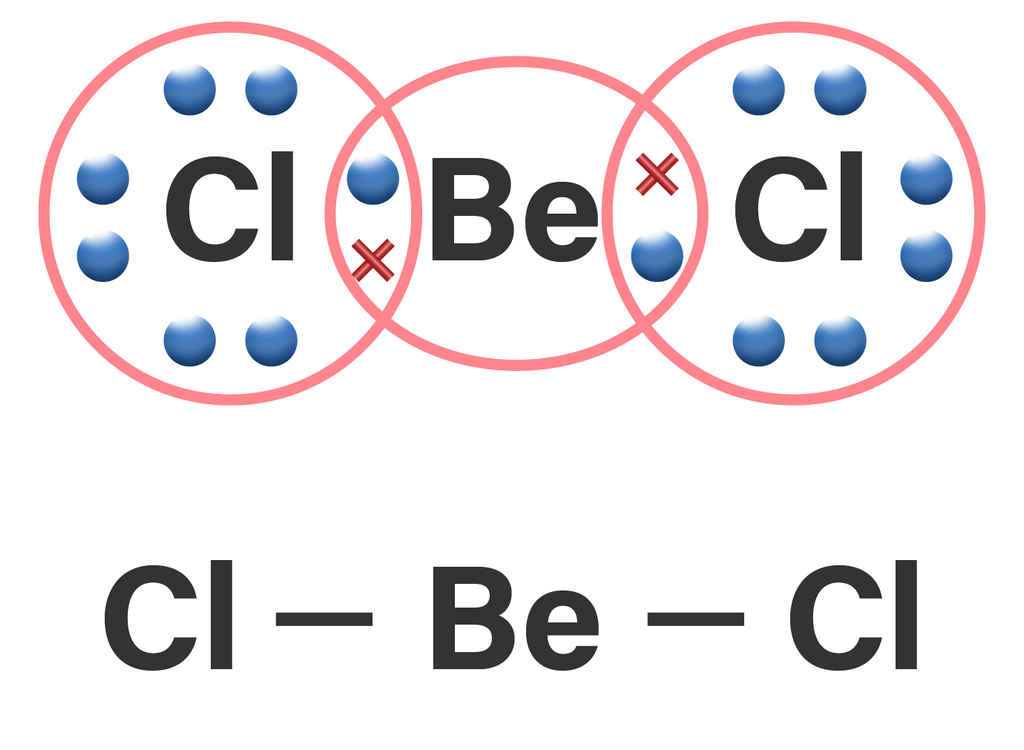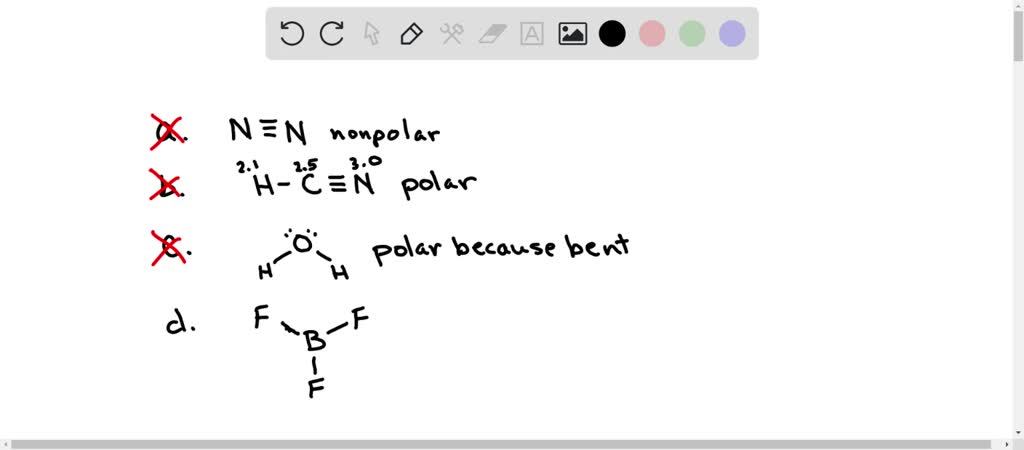
SOLVED: Which of the following statements concerning molecular polarity is correct? Select one: a. All diatomic molecules are polar: H2, N2, HF, and Cl2. b. All linear triatomic molecules are non-polar: CO2,
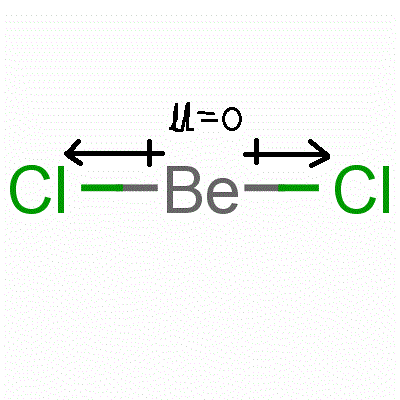
Be-Cl is polar but Becl2 is not why - Chemistry - Chemical Bonding and Molecular Structure - 9585317 | Meritnation.com

Is BeCl2 Polar or Nonpolar? - Polarity of Beryllium chloride | Molecular geometry, Covalent bonding, Electron configuration
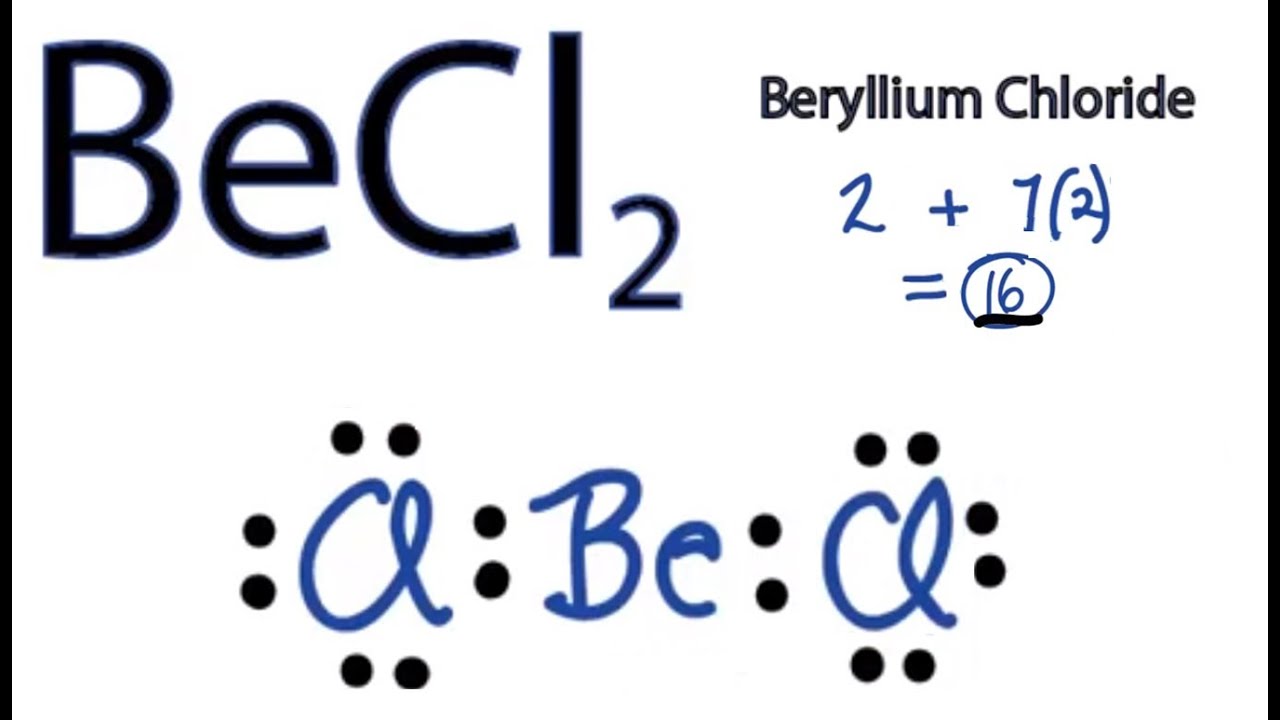






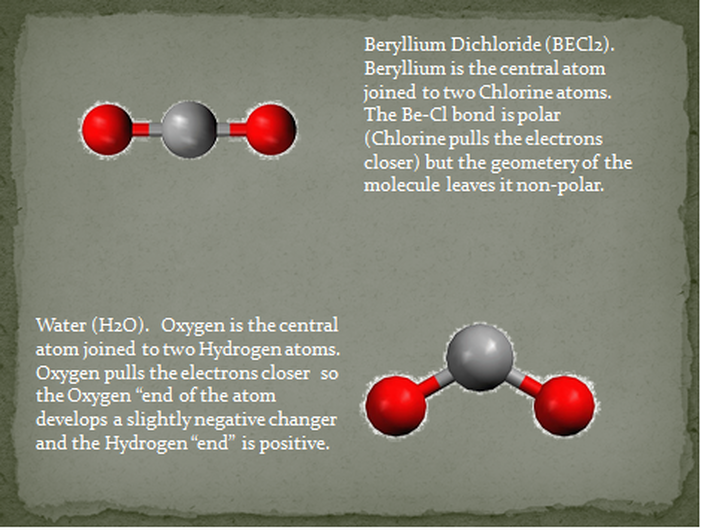


![Best Overview: Is BeCl2 Polar or Nonpolar [No#1] - Science Education and Tutorials Best Overview: Is BeCl2 Polar or Nonpolar [No#1] - Science Education and Tutorials](https://sciedutut.com/wp-content/uploads/2021/06/Hbr-cover-banner-.png)